Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ogawa, Shuichi*; Yoshigoe, Akitaka; Takakuwa, Yuji*
Vacuum and Surface Science, 62(6), p.350 - 355, 2019/06
Thermal oxidation of Si substrate is an indispensable process for the Si device fabrication. However, the influence of oxidation induced strain cannot be ignored for thin films. Synchrotron radiation real-time photoelectron spectroscopy was used as a method to measure simultaneously oxidation induced strain and oxidation rate. It was found that the acceleration of interfacial oxidation induced by thermal strain was observed for the rapid thermal oxidation. The results can be explained by the model in which point defects caused by strain become reaction sites at the SiO /Si interface.
/Si interface.
Ito, Hiroto*; Shiotsu, Hiroyuki; Tanaka, Yoichi*; Nishihara, Satomichi*; Sugiyama, Tomoyuki; Maruyama, Yu
JAEA-Data/Code 2018-012, 42 Pages, 2018/10
Chemical composition of fission products transported in nuclear facilities in severe accidents is controlled by slower chemical reaction rates, therefore, it could be different from that evaluated on the chemical equilibrium assumption. Hence, it is necessary to evaluate the chemical composition with reaction kinetics. On the other hand, databases applicable to the analysis of nuclear facilities have not been constructed because knowledge of reaction rates of complex chemical reactions in severe accidents is currently limited. Accordingly, we have developed the CHEMKEq code based on a partial mixed model with chemical equilibrium and reaction kinetics to decrease uncertainties of the chemical composition caused by the reaction rate. The CHEMKEq code, under mass conservation law, firstly evaluates chemical species obeying the chemical equilibrium model, and then, relatively slow reactions are solved by the reaction kinetics model. Moreover, the CHEMKEq code has a multiplicity of use in evaluations of chemical composition because general chemical equilibrium and reaction kinetics models are also available and databases required to calculation are external file formats. This report is the user's guide of the CHEMKEq code, showing models, solution methods, structure of the code and calculation examples. And information to run the CHEMKEq code is summarized in appendixes.
Suzudo, Tomoaki; Yamaguchi, Masatake
Journal of Nuclear Materials, 465, p.695 - 701, 2015/10
Times Cited Count:12 Percentile:70.51(Materials Science, Multidisciplinary)To investigate what atomic properties largely determine vulnerability to He embrittlement at grain boundaries (GB) of bcc transition metals, we introduced a computational model composed of first principles density functional theory and a He segregation rate theory model. Predictive calculations of He embrittlement at the first wall of the future DEMO fusion concept reactor indicated that variation in the He embrittlement is originated not only from He production rate related to neutron irradiation, but also from the He segregation energy at the GB that has a systematic trend in the periodic table.
 2) and (
2) and (

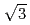 ) structures using surface X-ray diffraction
) structures using surface X-ray diffractionTamura, Kazuhisa; Mizuki, Junichiro
Journal of Physical Chemistry B, 109(26), p.12832 - 12836, 2005/07
Times Cited Count:2 Percentile:4.61(Chemistry, Physical)The kinetics of the phase transition between the (2  2) and (
2) and (

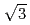 )-Bi structures on Au(111) was investigated using electrochemical methods and time-resolved surface X-ray diffraction. The temporal changes in the current value and the diffracted X-ray intensity that originated from the (2
)-Bi structures on Au(111) was investigated using electrochemical methods and time-resolved surface X-ray diffraction. The temporal changes in the current value and the diffracted X-ray intensity that originated from the (2  2)-Bi overlayer were monitored during the phase transitions at various overpotentials. For the (
2)-Bi overlayer were monitored during the phase transitions at various overpotentials. For the (

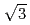 )
)  (2
(2  2) phase transition, the phase transition models determined by the X-ray and electrochemical measurements were a surface-diffusion controlled instantaneous nucleation-growth process and a Langmuir process, respectively. For the reverse transition, the phase transition models determined by X-ray and electrochemical measurements were a Langmuir adsorption process and a surface diffusion controlled nucleation-growth process, respectively.
2) phase transition, the phase transition models determined by the X-ray and electrochemical measurements were a surface-diffusion controlled instantaneous nucleation-growth process and a Langmuir process, respectively. For the reverse transition, the phase transition models determined by X-ray and electrochemical measurements were a Langmuir adsorption process and a surface diffusion controlled nucleation-growth process, respectively.
Yoshigoe, Akitaka; Moritani, Kosuke; Teraoka, Yuden
Shinku, 46(5), p.424 - 428, 2003/05
In order to study the reaction mechanism of the initial thermal Si(001) oxidation by O (1
(1 10
10 Pa) at the surface temperature region between 870K and 1120K, we have performed the
Pa) at the surface temperature region between 870K and 1120K, we have performed the 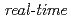 synchrotron radiation photoemission spectroscopy of Si-2p and O-1s levels. The oxygen uptake curves as a function of the oxygen exposure time were analyzed on the basis of kinetics. The Si oxidation states corresponding to the adsorbed oxygen amount were well clarified by the time evolution of Si-2p photoemission spectra.
synchrotron radiation photoemission spectroscopy of Si-2p and O-1s levels. The oxygen uptake curves as a function of the oxygen exposure time were analyzed on the basis of kinetics. The Si oxidation states corresponding to the adsorbed oxygen amount were well clarified by the time evolution of Si-2p photoemission spectra.
 ion irradiation
ion irradiationWakai, Eiichi; Ezawa, Tadashi*; Imamura, Junko*; Takenaka, Tsuyoshi*; Tanabe, Tetsuo*; Oshima, Ryuichiro*
Journal of Nuclear Materials, 307-311(Part.1), p.367 - 373, 2002/12
Times Cited Count:29 Percentile:84.88(Materials Science, Multidisciplinary)no abstracts in English
Nagano, Tetsushi
Gekkan Chikyu, 22(7), p.429 - 434, 2000/07
no abstracts in English
Arthur, R. C,*; Savage, D.*; Sasamoto, Hiroshi; Shibata, Masahiro; Yui, Mikazu
JNC TN8400 2000-005, 61 Pages, 2000/01
Kinetic data, including rate constants, reaction orders and activation energies, are compiled for 34 hydrolysis reactions involving feldspars, sheet silicates, zeolites, oxides, pyroxenes and amphiboles, and for similar reactions involving calcite and pyrite. The data are compatible with a rate law consistent with surface reaction control and transition-state theoly, which is incorporated in the geochemieal software package EQ3/6 and GWB. Kinetic data for the reactions noted above are strictly compatible with the transition-state rate law only under far-from-equilibrium conditions. It is possiblethat the data are conceptually consistent with this rate law under both far-from-equilibrium and near-to-equilibrium conditions, but this should be confirmed whenever possible through analysis of original experimental results, Due to limitations in the availability of kinetic data for mineral-water reactions, and in order to simplify evaluations of geochemical models of groundwater evolution, it is convenient to assume local-equilibrium in such models whenever possible. To assess whether this assumption is reasonable, a modeling approach accounting for coupled fluid flow and water-rock interaction is described that can be used to estimate spatial and temporal scale of local equiliblium. The approach is demonstrated for conditions involving groundwater flow in fractures at JNC's Kamaishi in-situ tests site, and is also used to estimate the travel time necessary for oxidizing surface waters to migrate to the level of a HLW repository in crystalline rock. The question of whether local equilibrium is a reasonable assumption must be addressed using an appropriate modeling approach. To be appropriate for conditions at the Kamaishi site using the modeling approach noted above, the fracture fill must closely approximate a porous medium, groundwater flow must be purely advective and diffusion of solutes across the fracture-host rock boundary must not occur. Moreover, the ...
Mukai, Satoru*; Kitao, Hideo*; Tachikawa, Hirokazu*; Fusaeda, Shigeki*; Yanagisawa, Ichiro*; Doi, Motoo*; *
PNC TJ1216 96-003, 106 Pages, 1996/03
None
Hosoi, Fumio; Omichi, Hideki; Akama, Kazuhiko*; Awai, K.*; Tokuyama, S.*; Sato, T.*
Nuclear Instruments and Methods in Physics Research B, 105(1-4), p.318 - 321, 1995/11
Times Cited Count:1 Percentile:23.89(Instruments & Instrumentation)no abstracts in English
 -radiation induced polymerization of unsaturated liposomes containing unsaturated lipid cholesterol, and saturated aliphatic acid
-radiation induced polymerization of unsaturated liposomes containing unsaturated lipid cholesterol, and saturated aliphatic acidHosoi, Fumio; Omichi, Hideki; Akama, Kazuhiko*; Tokuyama, S.*; Nakano, Y.*
JAERI-Conf 95-003, p.507 - 511, 1995/03
no abstracts in English
*; Yamamoto, Tadatoshi; ; Takebe, Shinichi; Ogawa, Hiromichi; Tanaka, Tadao; Mukai, Masayuki; Komiya, Tomokazu; S.Li*; Z.Wang*; et al.
JAERI-Research 94-009, 44 Pages, 1994/07
no abstracts in English
*; Tachimori, Shoichi; Naito, Shinjiro*
JAERI-M 93-056, 27 Pages, 1993/03
no abstracts in English
Hashimoto, Masashi; *; *; *
Radiochimica Acta, 63, p.173 - 177, 1993/00
no abstracts in English
; ; ; Kawakami, Waichiro
Kogai To Taisaku, 21(10), p.987 - 992, 1985/00
no abstracts in English
; ; ; ; ; Yoshida, Kenzo
EIM-82-111, p.27 - 36, 1982/00
no abstracts in English
 +C powders and compacts to UC
+C powders and compacts to UC
; ; ;
Journal of Nuclear Science and Technology, 19(3), p.222 - 230, 1982/00
Times Cited Count:12 Percentile:75.26(Nuclear Science & Technology)no abstracts in English
; ;
J.Appl.Polym.Sci., 26, p.2751 - 2762, 1981/00
Times Cited Count:0 Percentile:0.02(Polymer Science)no abstracts in English